Canine leishmaniosis is no longer just an “imported curiosity” for UK vets; recent data show a growing caseload in practices, making rapid, accurate diagnosis with tools like qPCR increasingly important for both individual dogs and wider disease control.
Canine leishmaniosis: why it matters in the UK
Over the past two decades, the UK has seen a steady rise in diagnosed Leishmania infantum infections, almost entirely in imported or travelled dogs, but with occasional non‑travel cases that raise concern about local transmission. A 2009 analysis of 257 confirmed cases entering the UK between 2005–2007 showed that most infected dogs had spent at least six months in Spain or other endemic areas, establishing imported dogs as the key reservoir.
More recent work has updated that picture: a 2026 study of UK practice data reported that imported animals remain the commonest source of diagnosed leishmaniosis, with cases clustering in southern England. A 2024 BSAVA summary of this work highlighted 368 dogs with leishmaniosis in UK practice records and documented an increased risk of recorded cases from 2017–2022 compared to 2014, particularly in neutered Pointing breeds and crossbreeds aged 3–6 years, with 34% of cases located in southeast England.
The UK Human Animal Infections and Risk Surveillance (HAIRS) group has also noted at least two autochthonous canine cases in non‑travel dogs, and the RVC has described suspected dog‑to‑dog transmission in a UK‑born dog, underscoring that under the right conditions, local maintenance is possible even without established sandfly vectors. For clinicians, this means that leishmaniosis should be on the differential list not only for the classic “Spanish rescue” but also for some UK‑raised dogs with compatible signs and relevant contact histories.
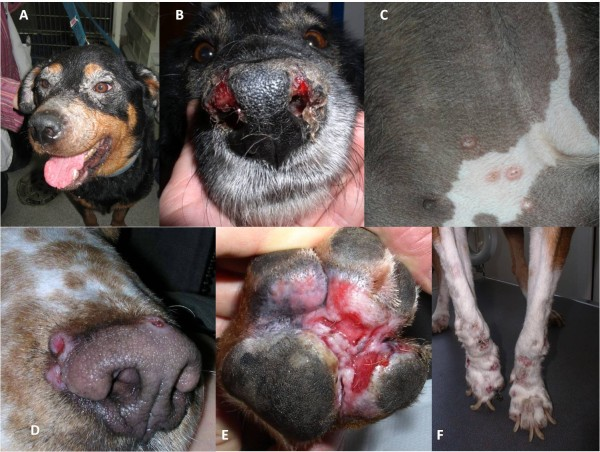
Different patterns of cutaneous lesions in CanL: A) Exfoliative periocular alopecia and blepharitis; B) Ulcerative nasal mucocutaneous lesions; C) Papular dermatitis in the inguinal region; D) Nodular crateriform lesions bordering the muzzle; E) Ulcerative erythematous lesions on the plantar surface of the paw and between pads; F) Onychogryphosis. (Taken from Solano-Gallego L et al., The LeishVet Group. LeishVet guidelines for the practical management of canine leishmaniosis. Parasit Vectors. 2011 May 20;4:86. doi: 10.1186/1756-3305-4-86. PMID: 21599936; PMCID: PMC3125381).
The diagnostic challenge: a stealthy parasite and imperfect tests
Diagnosing leishmaniosis is notoriously difficult because clinical signs are changeable and no single test is perfect. Typical features include weight loss, lymphadenopathy, dermatopathy, epistaxis and renal compromise; overlap with many other chronic diseases seen in UK practice, from immune‑mediated disease to chronic kidney disease and neoplasia.
The LeishVet guidelines emphasise a combined approach, using clinical staging alongside serology, cytology/histology and molecular tests to reach a robust diagnosis. However, a 2022 PLOS Neglected Tropical Diseases study that applied hierarchical modelling to canine leishmaniosis diagnostics showed just how imperfect individual assays can be: parasitological methods such as bone marrow microscopy and culture had sensitivities around 12–13%, rising to about 33% in some samples, while qPCR assays, though clearly superior, still missed a proportion of infected dogs and were vulnerable to low‑level contamination.
Crucially, that study highlighted that infection can be at least as common in apparently healthy dogs as in clinically sick ones, meaning the infection reservoir is larger and “quieter” than clinical caseload alone suggests. In a UK context, where the aim is to prevent establishment rather than simply manage endemic disease, this hidden reservoir makes early, accurate detection particularly valuable.
What recent UK data tell us about risk
Modern UK surveillance work has begun to unpack which dogs are most at risk and where they are being seen. In a recent analysis of practice records across the UK, nearly all cases with detailed histories appeared to have been infected before import, reinforcing the role of travel and rehoming from endemic regions as the dominant drivers.
The BSAVA‑reported study found that 39.7% of samples from at‑risk dogs tested seropositive, while 12% were PCR‑positive for L. infantum DNA, emphasising that a substantial minority of these dogs had detectable parasitaemia. Case mapping showed that the majority of dogs with leishmaniosis were seen in the southeast of England, aligning with travel and importation patterns as well as local demographic factors.
These findings dovetail with older work that documented imported cases from Spain and other Mediterranean countries, but they also extend it by quantifying risk factors such as neuter status, breed type and age, and by demonstrating that apparently healthy travelling dogs warrant consideration for screening. For UK practices, this evidence base supports a shift from “wait for classic signs” to proactive, risk‑based testing strategies.
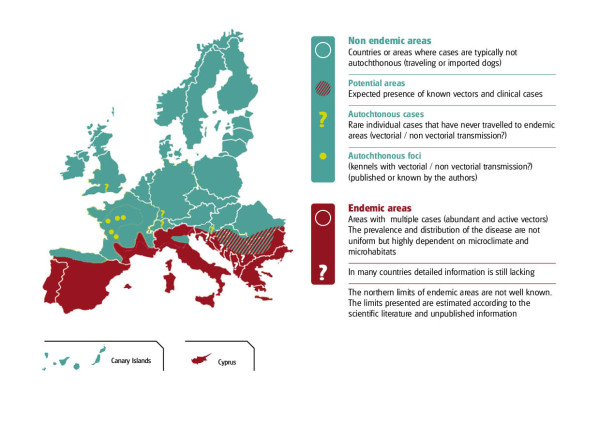
The distribution of canine L. infantum infection in Europe (taken from: Solano-Gallego L et al., The LeishVet Group. LeishVet guidelines for the practical management of canine leishmaniosis. Parasit Vectors. 2011 May 20;4:86. doi: 10.1186/1756-3305-4-86. PMID: 21599936; PMCID: PMC3125381).
Why rapid diagnosis is so important
Timely diagnosis matters in leishmaniosis for three main reasons: prognosis, iatrogenic risk and public/animal health. LeishVet‑based guidance and UK referral centre protocols consistently note that dogs diagnosed at earlier stages, before overt renal failure and severe immune‑complex disease, have better outcomes and respond more favourably to treatment combinations such as allopurinol plus leishmanicidal drugs. Waiting for azotaemia and marked proteinuria to develop not only worsens prognosis but also makes long‑term management more expensive and challenging for owners.
From a therapeutic and safety perspective, rapid confirmation or exclusion of leishmaniosis can prevent inappropriate high‑dose immunosuppression in a dog with vague systemic signs and hyperglobulinaemia, a scenario that risks exacerbating latent infection. Many UK clinicians will have encountered cases initially labelled as immune‑mediated disease or “idiopathic” chronic dermatopathy, later reclassified as leishmaniosis once more comprehensive testing was performed. Earlier molecular testing could shorten this diagnostic odyssey.
At a population level, prompt diagnosis feeds into screening before blood donation, travel, or rehoming, thereby reducing risk of transfusion‑associated or dog‑to‑dog transmission. The HAIRS assessment explicitly recognises that non‑vector routes such as transfusion, venereal spread and bite wounds can sustain infection chains in the absence of sandflies, so timely detection in donor and breeding animals is particularly important in the UK.
qPCR and lab‑on‑chip platforms: changing the game
Molecular methods, especially real‑time PCR, have transformed leishmaniosis diagnostics over the last decade, and recent work has sharpened our understanding of their strengths and limitations. A 2025 review of real‑time PCR techniques for leishmaniasis concluded that modern qPCR assays offer high specificity and good sensitivity across multiple sample types, with the added benefit of quantifying parasite load, which can inform staging and treatment monitoring.
In the hierarchical‑model framework discussed earlier, qPCR clearly out‑performed parasitological tests, with estimated sensitivities around 49–53% across sample types, compared with roughly 12–13% for bone marrow microscopy and culture. Specificity remained high (around 93–96%), although the authors detected a 4–5% rate of probable false positives due to low‑level qPCR plate contamination, highlighting the need for robust controls and workflow design. For UK labs handling relatively low volumes of leishmaniosis samples, these findings argue strongly for standardised protocols, internal controls and careful interpretation, but they also underline how much more powerful qPCR is than traditional methods.
A particularly relevant innovation is the emergence of portable, lab‑on‑chip real‑time PCR systems. A 2024 Journal of Clinical Microbiology study evaluated a qPCR for detection of L. infantum in canine samples, comparing it directly to a standard CFX96 real‑time system. In 173 extracted and 93 non‑extracted samples (blood, bone marrow, lymph node, buffy coat, conjunctival swabs and skin), the qPCR system achieved high analytical sensitivity and 100% clinical specificity, while bypassing conventional DNA extraction steps for certain sample types, which is also a feature of the UlfaQ In-clinic Real-time PCR – Portable Veterinary qPCR Device.
For practice and regional labs, the implications are significant: compact platforms capable of delivering near‑point‑of‑care qPCR with minimal infrastructure can dramatically shorten turnaround times and extend access to molecular testing beyond specialist centres and in to the practice. In settings where shipping to reference laboratories is the norm, same‑day results could become realistic, enabling clinicians to move rapidly from suspicion to evidence‑based management.
Bringing it together in UK practice
When you align the epidemiology with the diagnostic science, the case for rapid, qPCR‑enabled diagnosis in UK dogs becomes compelling. We now know that: imported and travelled dogs remain the primary reservoir; that apparently healthy animals can harbour infection; that clinical signs are non‑specific; and that slow or insensitive diagnostics delay recognition and control.
In practical terms, this supports risk‑based protocols such as: routine serology plus qPCR screening of imported or travelled dogs (especially those from Mediterranean rehoming organisations); prompt molecular testing in dogs with compatible but non‑specific signs; and use of rapid platforms to support pre‑donation and pre‑breeding screening. As in-clinic qPCR technologies mature, it is realistic to imagine a diagnostic ecosystem in which leishmaniosis testing is both fast and widely accessible across the UK, helping clinicians intervene earlier for individual dogs while contributing to national surveillance aimed at preventing the parasite from truly taking hold.
REFERENCES:
Kent SC, O’Neill DG, Stevens KB, Solano-Gallego L, Brodbelt DC. The frequency, risk factors and spatial distribution associated with having a diagnosis of leishmaniosis in dogs in the UK in 2019. PLoS One. 2026 Mar 4;21(3):e0341610. doi: 10.1371/journal.pone.0341610. PMID: 41779685; PMCID: PMC12959672.
Checa R, Sánchez-Vizcaíno F, Miró G, Pinchbeck G, Jones H, Noble P-J & Radford AD (2024) Updating the epidemiology of canine leishmaniosis in the United Kingdom through the use of electronic health data. Veterinary Parasitology. doi.org/10.1016/j.vetpar.2024.110350.
Human Animal Infections and Risk Surveillance (HAIRS) group. Qualitative assessment of the risk that canine leishmaniosis presents to the UK population. Accessed via: Human Animal Infections and Risk Surveillance (HAIRS) group: Qualitative assessment of the risk that canine leishmaniosis presents to the UK population January 2022
Shaw SE, Langton DA, Hillman TJ. Canine leishmaniosis in the United Kingdom: a zoonotic disease waiting for a vector? Vet Parasitol. 2009 Aug 26;163(4):281-5. doi: 10.1016/j.vetpar.2009.03.025. Epub 2009 Mar 26. PMID: 19369005.
Vital T, Teixeira AIP, Silva DM, de Carvalho BC, Dallago B, Hagström L, et al. (2022) Detecting Leishmania in dogs: A hierarchical-modeling approach to investigate the performance of parasitological and qPCR-based diagnostic procedures. PLoS Negl Trop Dis 16(12): e0011011. https://doi.org/10.1371/journal.pntd.0011011
Solano-Gallego L, Miró G, Koutinas A, Cardoso L, Pennisi MG, Ferrer L, Bourdeau P, Oliva G, Baneth G, The LeishVet Group. LeishVet guidelines for the practical management of canine leishmaniosis. Parasit Vectors. 2011 May 20;4:86. doi: 10.1186/1756-3305-4-86. PMID: 21599936; PMCID: PMC3125381.
Rihs JB, Vilela MT, Dos Santos JSC, Caldas S, Leite RS, Mol MPG. Exploring real-time PCR techniques for diagnosing leishmaniasis: key insights from a systematic review. Parasitol Res. 2025 May 21;124(5):54. doi: 10.1007/s00436-025-08503-2. PMID: 40397177; PMCID: PMC12095430.
RVC diagnoses first case of leishmaniosis transmission between dogs in the UK


